EVALUATION OF CRISPR/CAS9 GENOME-EDITING SYSTEM IN HUMAN STEM CELLS HSCS: THERAPEUTICS AND DIAGNOSTICS PROSPECTS
DOI:
https://doi.org/10.71000/ce0xek27Keywords:
CRISPR/Cas9, gene editing, genetic therapy, hematologic diseases, human stem cells, regenerative medicine, sickle cell anemiaAbstract
Background: CRISPR/Cas9 genome-editing technology has revolutionized human stem cell (HSC) research, offering novel therapeutic and diagnostic applications. HSCs play a crucial role in regenerative medicine and genetic therapies due to their ability to self-renew and differentiate into various blood cell lineages. The precise genome-editing capability of CRISPR/Cas9 allows for targeted gene modifications, enabling the correction of inherited disorders, disease modeling and the discovery of novel biomarkers. Despite significant advancements, challenges such as off-target effects, delivery efficiency, and ethical concerns persist, requiring further research and optimization.
Objective: This systematic review evaluates the therapeutic and diagnostic potential of CRISPR/Cas9 genome editing in HSCs, focusing on its efficacy in gene correction for hematologic disorders, disease modeling and biomarker discovery.
Methods: A systematic review was conducted following PRISMA guidelines, analyzing studies published between 2015 and 2024. Literature searches were performed in PubMed, Web of Science, and Scopus using MeSH-aligned keywords. The inclusion criteria encompassed peer-reviewed studies utilizing CRISPR/Cas9 for gene modification in HSCs for therapeutic and diagnostic applications. Exclusion criteria included studies that lacked experimental validation, did not focus on HSCs, or were non-English publications. Out of 85 initially retrieved studies, 40 met the inclusion criteria, and 15 were selected for final synthesis.
Results: CRISPR/Cas9 gene-editing strategies in HSCs were categorized as gene knockout (53%), gene activation (40%), and dual knockout/activation (7%). Hematological disorders, including sickle cell anemia and beta-thalassemia, accounted for 35% of studies, demonstrating up to 90% correction in β-globin mutations. Neurodegenerative diseases constituted 20% of studies, where knockout of amyloid precursor protein (APP) in Alzheimer’s models resulted in a 60% reduction in plaque accumulation. Muscular dystrophy studies (10%) showed 75% improvement in dystrophin expression through gene activation. High-throughput CRISPR screening was employed in 15% of studies for biomarker identification. Despite promising outcomes, off-target mutations were observed in 28% of studies, and viral vector-based delivery methods were used in 65%, raising safety concerns.
Conclusion: CRISPR/Cas9 genome editing in HSCs presents a ground-breaking approach for treating genetic disorders and enhancing precision medicine. Its potential to correct disease-causing mutations, model complex disorders, and identify novel therapeutic targets is substantial. However, challenges in delivery methods, long-term safety, and ethical considerations remain barriers to clinical translation. Future research should focus on optimizing high-fidelity Cas9 variants, improving non-viral delivery methods, and addressing ethical concerns to ensure the safe and effective application of CRISPR/Cas9 in regenerative medicine.
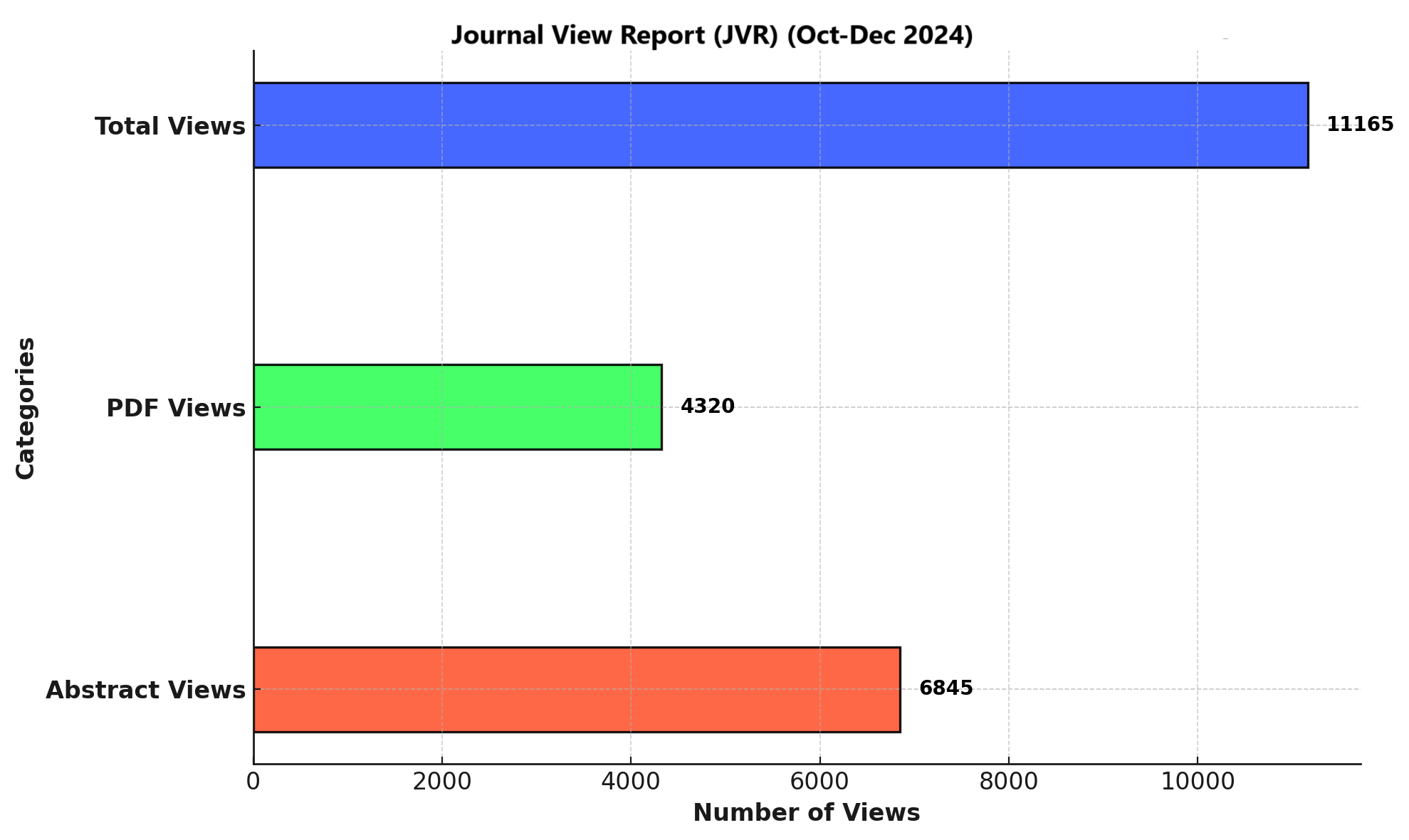
Downloads
Published
Issue
Section
License
Copyright (c) 2025 Ehsan Ul Haq, Haseeb Khaliq, Ayesha Muddasser, Misha Aslam, Farwa Zafar, Naveera Mazhar, Amna Naheed Khan (Author)

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.